-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

3M 90021 - DRESSING TEGADERM HYDROCOLLOID OVAL 10/BX 10BX/CA
3M Tegaderm Hydrocolloid Thin Dressing(Oval), 2.75 in. x 3.5 in., 7 cm x 9 cm, 10 cm x 12 cm
An advanced hydrocolloid dressing for low to moderately exuding wounds
3M Tegaderm Hydrocolloid Thin Dressing are sterile wound dressings, which consist of a hypoallergenic, hydrocolloid adhesive with an outer clear adhesive cover film. The film is moisture vapor permeable, waterproof, and impermeable to liquids, bacteria, and viruses*. The products are not designed, sold or intended for use except as indicated.
- May be worn up to 7 days.
- Conformable dressing is easy to apply.
- Dressing maintains moist wound environment for enhanced healing.
- Outer film is 3M Tegaderm Transparent Film Dressing technology which applies easily and eliminates need for taping edges, is easy to clean, minimises exudate leakage and is impermeable to liquids, bacteria and viruses.
- For minimally draining wounds.
7cm x 9cm Thin Hydrocolloid Dressing, Oval, Film border, Designed for long wear time, Overall size 10cm x 12cm
Reasons for using Tegaderm Hydrocolloid Thin Dressing
1. Unique Tegaderm Film border provides:
- Average wear time of almost 7 days
- A waterproof backing and protection from bacteria and viruses
2. Available in - square and oval dressings
3. Transparent
4. Does not contain - gelatin, pectin or tackifiers
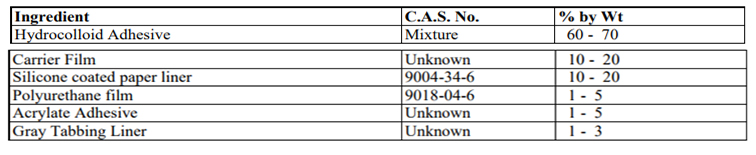
Composition/information on ingredients
Indications for Use
Tegaderm Hydrocolloid Thin Dressing is indicated for partial thickness dermal ulcers, leg ulcers, superficial wounds, abrasions, first- and second-degree burns, and donor sites. It may also be used as a protective dressing on at-risk, undamaged skin or on skin beginning to show signs of damage from friction or shear.
Suggested Applications
- Superficial, dry to lightly exudating partial thickness dermal ulcers and skin tears
- May be used as a protective covering over fragile skin or newly healed skin
- Donor sites
Key Benefits
Clinically Effective:
- Unique hydrocolloid adhesive rapidly absorbs exudate without breaking down or leaving residue in the wound for easier dressing application.
- Dressing ensures an optimal moist wound healing environment which has been shown to enhance healing.
- Smooth outer surface protects skin from shearing caused by friction, may be used for prevention of damage to intact skin.
- May be worn up to seven days.
Protection from Contaminants:
- Outer film barrier protects the wound and surrounding skin from contaminants and body fluids.*
- Provides a consistently high moisture vapour transmission to reduce the potential for periwound skin maceration.
*Laboratory testing has proven that the film provides a barrier against HIV-1 and HBV while the dressing remains intact without leakage.
Patient Benefits
Tegaderm Hydrocolloid Dressing provides excellent patient comfort, dressing adherence and wound management. The thin, flexible, conformable, low-profile dressings are comfortable because there is less bulk to catch on bed linens. With film dressing border, there is less likelihood of edge lift. Water resistance allows the patient to shower or have a bed bath without wound contamination.
Unique Features
- Special film bordered "notch" area allows application in close proximity to the anal opening while still providing a barrier to contamination by feces or urine.
- Provides non-tenting border application into the gluteal cleft area to seal against incontinence and other body fluids.
- Leaves no hydrocolloid residue in the wound for fast, easier dressing change.
- Outer film layer provides consistently high moisture vapour transmission reducing the potential for periwound skin maceration.
- Film border provides secure seal against outside contaminants.
The high rate of moisture vapour transmission reduces the potential for maceration. The barrier cover plus absorbency promote long wear time. For use on partial and full thickness dermal ulcers, superficial wounds, abrasions, superficial partial-thickness burns and donor sites.
3M Tegaderm Hydrocolloid Dressing Stays in Place for Long Wear Time and Potential Cost Savings
Ideal for Low- to Moderate-Draining Wounds
- Proven up to 7 day wear time provides undisturbed moist wound environment which has been shown to enhance healing
- Smooth outer surface helps reduce the risk of shear and friction
- Adhesive dressings continue to adhere and protect the wound in the presence of incontinence
- Long wear time means less dressing changes and potentially lower costs
Maximize Sacral Protection
- The sacral shape stays in place
- Provides non-tenting border application in the gluteal cleft to seal against incontinence
- Easy to wipe off after incontinent episodes
- Reduced need for unnecessary dressing changes
- Convenient one-handed application
Variety of Shapes and Sizes
Tegaderm Hydrocolloid Dressings are available in three convenient shapes - oval, sacral and square. Oval and sacral shapes have a film border. The film border eliminates the need for tape and makes dressing application easier. Ovals and squares are also available in a thin dressing design.
Comfort for Patients
Tegaderm Hydrocolloid Dressings provide excellent patient comfort, dressing adherence and wound management. The flexible, conformable, low-profile dressings are comfortable because there is less bulk to catch on bed linens. With film dressing border, there is less likelihood of edge lift. Water resistance allows the patient to shower or to have a bed bath.

Clinically Proven
Three separate lots (6 replicates for each lot) of 3M Tegaderm Hydrocolloid Dressing and DuoDERM CGF Dressing were tested for absorbency. Results were recorded as grams of solution absorbed per gram of dressing material. Absorbency was measured at 2, 4, 24 and 48 hours. The results from the absorbency testing indicate that Tegaderm Hydrocolloid Dressing had a statistically significant higher mean absorbency than DuoDERM CGF Dressing at every time point (p

*In vitro testing shows that the transparent film of Tegaderm , Tegaderm Advanced, Tegaderm HP and Tegaderm CHG dressings provides a viral barrier from viruses 27 nm in diameter or larger while the dressing remains intact without leakage.
Commonly Asked Questions
When would you select a thin hydrocolloid dressing?
Answer: Tegaderm Thin Hydrocolloid is intended for using as a dressing for superficial, mildly exuding partial thickness dermal wounds and skin tears.
Question: Is the dressing waterproof?
Answer: Yes. The breathable outer film layer provides a protective cover and is impermeable to liquids, bacteria and viruses*.
Question: Can you cut the dressing?
Answer: Yes. The dressing may be cut and, when overlapped, will self seal. Taping the cut edge will protect from premature lifting or edge roll.
Precautions
- Treatment of any skin ulcer should be part of a well-defined plan for ulcer management and under the supervision of a health care professional.
- When using Tegaderm Hydrocolloid Dressings, the wound initially appears larger in size and depth as unnecessary tissue is cleaned away. This increase should be accompanied by an improved appearance of the wound. If the wound gets larger after the first few dressing changes, see a health care professional.
- Observe the wound for signs of infection. If infection occurs, the signs of which may be fever, increased pain, redness, bleeding, swelling, or an unusual odor or discharge see a health care professional. Tegaderm Hydrocolloid Dressings may be used on infected wounds only under the care of a health care professional.
- Rarely, irritation (reddening, inflammation), maceration (whitening of the skin), or hypergranulation (excessive tissue formation) may develop in some wounds under hydrocolloid dressings. Should these occur, consult a health care professional.
- If the wound does not begin to show signs of healing or if any other unexpected symptoms occur, consult a health care professional.
Directions for Use Before Using the Dressing
- Clip excess hair for patient comfort.
- Clean the skin and the wound thoroughly.
- Allow the skin to dry before applying the dressing.
- If the periwound skin is fragile or exposure to drainage is likely, apply 3M Cavilon No Sting Barrier Film to the periwound skin.
- Evaluate the wound and select the correct dressing size so that 2.5 cm (1 inch) of the dressing, including the adhesive border, extends beyond the wound edge.
Applying the Dressing
Remove the paper liner from the dressing by lifting and pulling one of the square end tabs marked "1", exposing the adhesive surface (Figure 1). Minimize contact with the border or the adhesive (bottom) side of the dressing. 2.Center the dressing over the wound. Gently press the adhesive (bottom) side down, and press from the center outward. Avoid stretching the dressing or the skin (Figure 2). 3.Press down and smooth the film edges to ensure good adhesion (Figure 3). 4.Remove the top delivery film by lifting and pulling one of the center tabs marked "2" toward the edge of the dressing (Figure 4). Smooth down the dressing edges as you are removing the film. Remove the other side of the top film in the same way. 5.Gently tear off the square end tabs marked "1" at the perforations in a downward direction and discard (Figure 5). Avoid lifting the film edge while removing the tabs. Secure the entire film edge by pressing firmly. |
|
Applying the Dressing to Heels or Elbows
1.Slit the oval dressing from one tab end to the center (Figure 6). 2.Secure the uncut half of the dressing to the bottom of the heel or elbow (Figure 7). 3.Secure one half of the cut side of the dressing to the upper heel or the upper elbow (Figure 8). 4.Remove the top delivery film and tab from that part of the dressing on the upper heel or elbow (Figure 9). 5.Secure the remaining half of the cut side of the dressing by overlapping it on the secured portion of the dressing (Figure 10). Remove the remaining top delivery film and the tabs. 6.Secure the cut edges by pressing firmly (Figure 11). |  |
Removing the Dressing
1.Carefully lift the film edges from the skin. If there is difficulty lifting the dressing, apply tape to the edge and use the tape to lift.
2.Continue lifting the film until all edges are free from the skin surface.
3.Remove the dressing slowly, folding it over itself. Pull carefully in the direction of hair growth.
Note: It is not unusual for wounds to have an odor. This may be noticed when the dressing is removed or when leakage occurs. The odor should disappear after the wound is cleaned.
Specifications
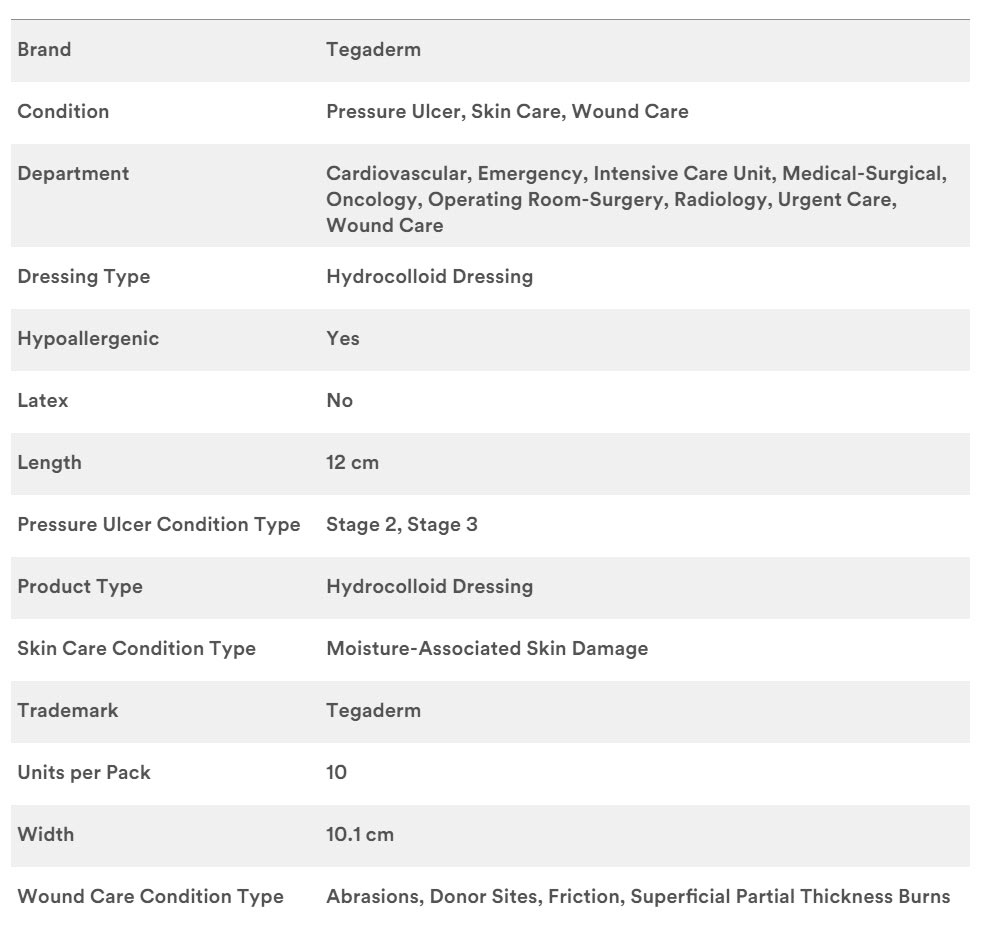

3M #90021, DRESSING, TEGADERM THIN, OVAL 4"X 4.75", EACH
$3.75 per EACH

3M #90021, Dressing Tegaderm Island Oval 4x4-3/4" Abs Adh Adhr 10/Bx, 10 BX/CA
$437.90 per CASE

3M #90021, Dressing Tegaderm Island Oval 4x4-3/4" Abs Adh Adhr 10/Bx, 10 BX/CA
$502.92 per CASE

3M #90021, Tegasorb 7x9cm Thin 4X4-3/4" Ovl 10/BX, 10 BX/CS
$43.79 per BOX



