-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Avanos Medical 0120-12-0.8 - Mic-Key Low-Profile Gastrostomy Feeding Tube - 12 FR, 0.8 CM, Each
 |  |
Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube
The AVANOS MIC-KEY Low-Profile Gastrostomy Feeding Tube allows for delivery of enteral nutrition and medication directly into the stomach and/or gastric decompression. MIC-KEY Low-Profile Gastrostomy Feeding Tube with SECUR-LOK (CE) is a step up from conventional gastrostomy feeding tubes. Highly popular and widely prescribed, the MIC-KEY feeding tube is unobtrusive and easy to conceal, making it an ideal feeding tube for virtually all individuals. The MIC-KEY Low-Profile Gastrostomy Feeding Tube features a high clarity medical grade silicone construction, designed for visibility and drapability. The design also features an inflatable internal retention balloon, as well as a patented universal connector designed to preserve tube life and minimize unintentional disconnects.
Quality Build for Quality Results. Avanos Medical, a leader in the enteral feeding market, offers a wide variety of innovative, high-quality enteral feeding tubes and accessories uniquely designed for delivering nutrition and medication to pediatrics and adults. As the pioneer in the development of the first balloon retained tube designed for gastrostomy feeding, Avanos Medical continues to manufacture tubes and accessories with the highest standards of workmanship at Avanos Medical state-of-the-art manufacturing facilities. Avanos Medical products provide consistent performance, and are designed for safety and convenience.
Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube Features & Benefits
- Sits at skin level
- Easy to conceal
- Limited interference with clothing
- Various sizes for adults
- Enables patients to enjoy life to the fullest
- Medical grade silicone construction
- Low-profile design
- Recessed distal tip design
- Proximal anti-reflux valve
About The Mic-Key G-Tube
A MIC-KEY low-profile gastrostomy feeding tube (MIC-KEY G-tube) has been inserted into your stomach through the abdominal wall. There is an inflatable balloon at one end and an external base or external retention disc at the other. This tube allows the intake of food and water that your body requires. Your medical professional has measured your stoma length including the optimal diameter (French size or Charrire) to ensure that you have the right size MIC-KEY G-tube.

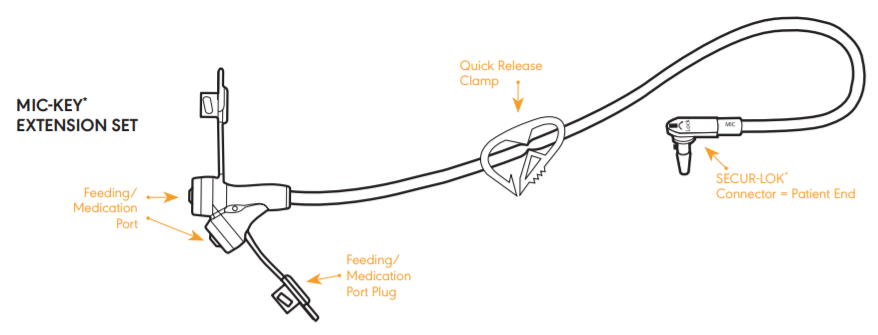
Mic-Key Extension Set
Use this extension set for continuous feeding. To attach the extension set, align the black line on the patient end of the extension set with black line on the feeding port of the tube. Insert the "nose" of the SECUR-LOK connector into the feeding port and rotate it one quarter turn clockwise. Open the extension set feeding port and screw the ENFit connector of the feeding set to the extension set. The extension set "swivels" with movement and allows you to change position during feeding.
Wash the extension set after every feeding with warm soapy water and rinse it thoroughly. Prompt flushing and rinsing prevents the formula from drying and building up. For hygienic and functionality reasons it's recommended to replace the MIC-KEY Extension Sets at least every 2 weeks.
The External Base of Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube
The external base or retention disc holds the tube in place yet allows air circulation to the skin around and underneath it. The bottom of the base should rest just above the skin surface. A good fit is considered 3 mm above the skin. The external base consists of 3 parts: the feeding port, closure cap and the balloon port.

The Feeding Port of Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube
An anti-reflux valve is located inside the feeding port. This helps prevent stomach contents from leaking out of the tube once the closure cap is opened. Once the patient end of the extension set is connected to the tube, the anti-reflux valve will open, allowing the administration of tube feeds or medication or the aspiration of gastric contents. The extension set can also be used for venting. This is also called decompression or burping. Never try to feed or put medication directly into the feeding port without using an extension set.
It is important to keep the feeding port and anti-reflux valve clean. Dried tube feeds may lodge inside the recess and hold the valve open. The best preventative measure is to flush thoroughly with enough water through the extension set to clear all tube feeds and to use cotton tipped applicators and water to clean the valve. Be sure that residual tube feeds is not left to pool and dry inside the valve opening.
Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube Balloon Valve
Your feeding tube has a balloon inside the stomach that has been inflated to hold the tube in place. Your specialist filled it with water when the tube was inserted. The balloon is inflated and deflated by inserting a luer slip syringe into the balloon insufflation port (balloon valve). It should only be used when checking the balloon volume or replacing the MIC-KEY G-tube. It is important to never attempt to feed through the balloon insufflation port. It is also important to keep this port clean. The recess in the port can trap foreign material and it must be clean to function properly.
Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube - Indications for Use
The AVANOS MIC-KEY Low-Profile Gastrostomy Feeding Tube is indicated for use in patients who require long-term feeding, are unable to tolerate oral feeding, who are at low risk for aspiration, require gastric decompression and/or medication delivery directly into the stomach.
Contraindications of Avanos 0120-12-0.8 Mic-Key Low-Profile Gastrostomy Feeding Tube
Contraindications for placement of a low-profile gastrostomy feeding tube include but are not limited to ascites, colonic interposition, portal hypertension, peritonitis, and morbid obesity.
Avanos MIC-KEY Low-Profile Gastrostomy Feeding Tube 0120-12-0.8 Device Characteristics
| What MRI safety information does the labeling contain? | MR Conditional |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437): | No |
| Device labeled as ""Not made with natural rubber latex"": | No |
| For Single-Use: | Yes |
| Prescription Use (Rx): | No |
| Over the Counter (OTC): | No |
| Kit: | No |
| Combination Product: | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P): | No |

Avanos Medical #0120-16-0.8, Mic-Key Low-Profile Gastrostomy Feeding Tube - 16 FR, 0.8 CM, Each
$173.27 EACH

Avanos Medical #0120-20-0.8, Mic-Key Low-Profile Gastrostomy Feeding Tube - 20 FR, 0.8 CM, Each
$173.27 EACH

Avanos Medical #0120-12-3.5, Mic-Key Low-Profile Gastrostomy Feeding Tube - 12 FR, 3.5 CM, Each
$173.27 EACH

Avanos Medical #0120-12-4.0, Mic-Key Low-Profile Gastrostomy Feeding Tube - 12 FR, 4.0 CM, Each
$173.27 EACH

