-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Carefusion 260100 - Chloraprep Swabstick, 1.75 mL, Single-Use Antiseptic Applicator, 480/CS
The CareFusion 260100 Chloraprep Swabstick is a single-use antiseptic applicator designed for preoperative and procedural skin preparation. It contains 1.75 mL of Chlorhexidine Gluconate (CHG) and Isopropyl Alcohol (IPA), an effective combination that rapidly eliminates a broad spectrum of bacteria and provides long-lasting antimicrobial protection. The applicator is designed to ensure uniform application with minimal dripping, enhancing patient safety and procedural efficiency.
This swabstick is widely used in surgical, procedural, and healthcare environments where maintaining a sterile field is essential.
 |  |
Key Benefits
- Effective Antimicrobial Action The combination of CHG and IPA rapidly kills bacteria and reduces the risk of infections.
- Long-Lasting Protection Provides extended antimicrobial activity, ensuring a cleaner surgical or procedural site.
- Single-Use, Sterile Design Reduces cross-contamination risks and enhances patient safety.
- Easy Application The swabstick design allows controlled application without excess dripping.
- Broad Clinical Utility Suitable for preoperative skin preparation, IV insertions, and catheter placements.
Compatibility and Applications
The CareFusion 260100 Chloraprep Swabstick is compatible with standard preoperative and procedural skin preparation protocols. It is widely used in hospitals, outpatient clinics, and surgical centers.
Common application scenarios include:
- Surgical Skin Preparation Applied before surgical procedures to reduce infection risks.
- IV Site Preparation Used to disinfect skin before IV insertions.
- Catheter Insertion Sites Reduces microbial presence for safer catheter placements.
- Minor Medical Procedures Ideal for pre-procedural skin antisepsis.
Application instructions
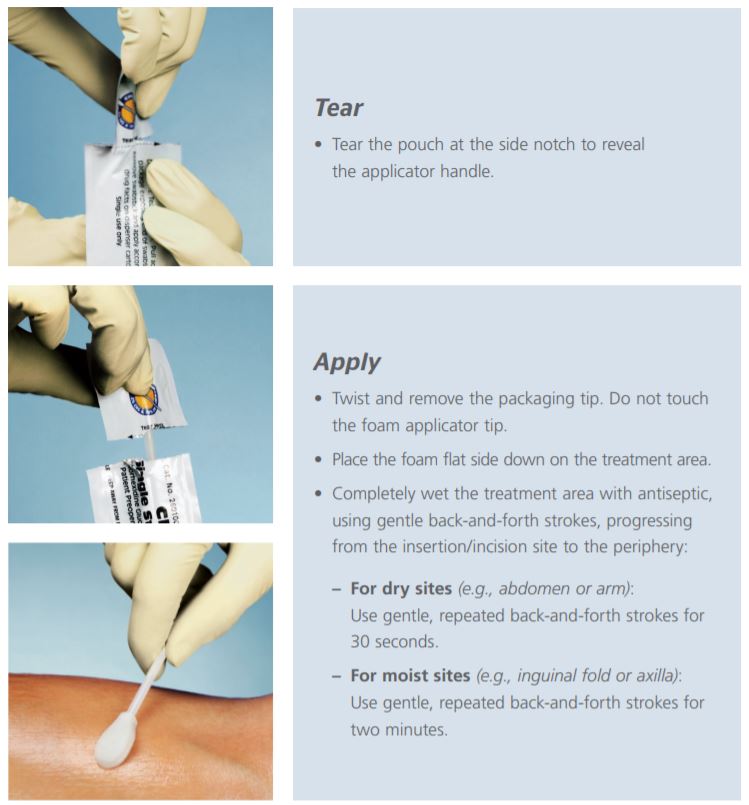
Before using the ChloraPrep swabstick applicator, read the instructions on the package. Use in accordance with the policies and procedures of your hospital.

Usage Guidelines
To ensure optimal performance and safety, follow these best practices:
- Open the sterile packaging just before use.
- Apply the swabstick using a back-and-forth motion for effective antimicrobial coverage.
- Allow the site to dry completely before proceeding with any surgical or procedural steps.
- Dispose of the swabstick in accordance with facility guidelines for medical waste.
- Follow the manufacturers instructions for detailed procedural guidelines.
Certifications, Safety Standards, and Regulatory Approvals
The CareFusion 260100 Chloraprep Swabstick meets stringent regulatory requirements, ensuring compliance with global medical safety standards:
- FDA Approved Certified for medical use in the United States, ensuring adherence to strict quality and safety standards.
- CE Marked Compliant with European health, safety, and environmental protection regulations.
- ISO 13485 Certified Manufactured under international quality management standards for medical devices.
Comparison with Competitor Products
Compared to other preoperative antiseptic applicators, the CareFusion 260100 offers:
- Faster bacterial kill time compared to standard alcohol-based preps.
- Extended antimicrobial activity, reducing the risk of post-procedural infections.
- Superior application control, minimizing excess fluid runoff and wastage.
- Single-use sterility, ensuring consistent aseptic technique.
Typical User Profile and Application Scenarios
The CareFusion 260100 Chloraprep Swabstick is primarily used by:
- Surgeons and OR Staff For preoperative skin preparation before surgical procedures.
- Nurses and IV Specialists Ensuring antiseptic skin preparation before IV placements.
- Catheterization Teams Reducing infection risk before catheter insertions.
- Emergency and Ambulatory Care Staff Providing rapid skin antisepsis in urgent care settings.
Best Practices for Use and Maintenance
- Store in a dry, temperature-controlled environment to maintain sterility.
- Ensure proper handling with gloved hands to prevent contamination.
- Dispose of after single use in accordance with medical waste regulations.
- Train healthcare staff on proper antiseptic application techniques to maximize efficacy.
Regulatory and Safety Information
This product complies with global safety and regulatory standards to ensure medical reliability. Users should refer to their facilitys guidelines for antiseptic skin preparation and follow appropriate infection control protocols.
How to Order
To purchase the CareFusion 260100 Chloraprep Swabstick, add the product to your cart and proceed to checkout. For bulk orders or additional inquiries, contact CIA Medical customer service at (312) 275-5850 or submit a request via the contact form on this page.

