-
Catheters (6,800+)
- Angiocatheters (50+)
- Closed System Catheters (300+)
- External Catheters (620+)
- Hydrophilic Catheters (140+)
- IV Catheters (1,200+)
- Non-Hydrophilic (20+)
- Plastic Catheters (200+)
- Rubber Catheters (700+)
- Silicone Catheters (770+)
- Ureteral Catheters (100+)
- Urethral Catheters (450+)
- Venous Catheters (240+)
-
Coronavirus (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (20,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Custom Kits
- Dental (14,000+)
- Gloves (8,000+)
-
Gynecology & Urology (1,000+)
- Bed Side Drainage Bags (350+)
- Circumcision (150+)
- Cord Clamps and Clippers (60+)
- Disposable Vaginal Specula (60+)
- Enema Bags (30+)
- External Catheters (620+)
- Foley Catheters and Trays (1,200+)
- Identification (1100+)
- Leg Bag Accessories (10+)
- Leg Bags (280+)
- Reusable Vaginal Specula (900+)
- Specimen Collection (200+)
- Tubing & Connectors (17,000+)
- Urinals / Bed Pans (1,300+)
- Urine Collectors (60+)
- Urological Irrigation Products (10+)
- Vaginal Specula Illumination (2+)
- Systems (11,000+)
- Hygiene (1,000+)
- Incontinence (1,000+)
-
Infection Control (2,500+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Iodine (460+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Infusion All (2,000+)
- IV Bags - Empty (300+)
- IV Bags - Filled (100+)
- Masks (3,800+)
-
Medical Apparel (23,000+)
- Arm Sleeves (240+)
- Beard Covers (20+)
- Bouffant Caps (200+)
- Compression Socks (80+)
- Coveralls (500+)
- Disposables (100+)
- Isolation Gowns (360+)
- Lab Coats (2,200+)
- Lab Jackets (300+)
- Patient Gowns (300+)
- Procedural Gowns (230+)
- Scrubs (20,000+)
- Shoe Covers (270+)
- Surgeon Caps (40+)
- Surgical Gowns (70+)
- Surgical Hoods (20+)
- Surgical Masks (330+)
- Ostomy (400+)
-
PPE (20,000+)
- Bacterial Filters (170+)
- Bleach (360+)
- Coveralls (500+)
- Disinfectant Wipes (350+)
- Face Shields (200+)
- Gloves (8,000+)
- Gowns (2,300+)
- Isopropyl Alcohol (170+)
- IV Therapy (2,000+)
- Masks (3,700+)
- Pulse Oximeters (250+)
- Sanitizer (670+)
- Scrubs (23,000+)
- Soap (1,500+)
- Stethoscopes (700+)
- Thermometers (950+)
- Respiratory (500+)
- Sanitizer (600+)
- Surgical Supplies (14,000+)
- Sutures (7,500+)
- Syringes & Needles (14,000+)
-
Wound Care (5,000+)
- ABD Pads (100+)
- Adhesive Bandages (650+)
- Advanced Wound Care (400+)
- Applicators (6,700+)
- Burn care (240+)
- Dressings (7,500+)
- Elastic Bandages (1,600+)
- Gauze (3,300+)
- Ice / Heat Packs (280+)
- Medical Tape (820+)
- Non-Adhering Dressings (100+)
- Ointment & Solutions (450+)
- Self-Adherent Wraps (200+)
- Sponges (2,400+)
- Staple & Suture Removal (1,500+)
- Tegaderm (450+)
- Transparent Dressing (800+)
- Wound Care Prep (120+)
- Wound Cleansers (100+)
- Sales & Deals (100+)
- 3M (4,200+)
- Alaris Medical (600+)
- Amsino International (550+)
- Avanos Medical (40+)
- B Braun (1,500+)
- Baxter (750+)
- BD (2,800+)
- BSN Medical (2,000+)
- Cables & Sensors (3,200+)
- C.R. Bard (4,200+)
- Cardinal Health (6,800+)
- CareFusion (2,100+)
- ConMed (1,500+)
- Cook Medical (600+)
- Covidien (9,500+)
- DeRoyal (6,000+)
- Dukal (1,300+)
- Ethicon (4,100+)
- GE Healthcare (1,000+)
- Hartmann (600+)
- Hospira (530+)
- ICU Medical (1,700+)
- Masimo (170+)
- Medline (54,000+)
- Midmark (2,500+)
- Roche (300+)
- Smiths Medical (4,000+)
- Sunset Healthcare (450+)
- TrueCare Biomedix (20+)
- View All Brands (5,000+)

Covidien SMM5043 - Suture Maxon Mono Clr P12 4-0 18" 36/BX

Maxon Monofilament Absorbable Sutures
Maxon synthetic absorbable sutures are prepared from polyglyconate, a copolymer of glycolic acid and trimethylene carbonate.
Maxon reverse cutting needles feature tri-coined, reverse cutting edges for use in dense connective tissue and skin. The cutting edge is on the convex surface of the needle arc, facilitating needle passage while minimizing tissue cut-through. Premium reverse cutting needles are made exclusively of Surgalloy material for added strength and ductility.
Premium Reverse Cutting - Maxon 4-0 Clear 18" P-12
| Item Number | Structure | Suture Type | Absorption Profile | Size USP | EP | Length. (CM) | Color | Box Qty |
| SMM5043 | Monofilament | Long-Term Absorbable | 180 Days | 4-0 | 1.5 | 45 | Clear | 36 |
Strength and Security for Extended Wound Healing
Maxon synthetic absorbable sutures are indicated for use in general soft tissue approximation and/or ligation, including use in pediatric cardiovascular tissue, where growth is expected to occur, and in peripheral vascular surgery. They are not indicated for use in adult cardiovascular tissue, ophthalmic surgery, microsurgery and neural tissues.
The advanced extrusion process of the molecule of polyglyconate gives the suture:
- Excellent in-vivo strength retention
- Excellent knot tying security
- Minimal memory
The Strength and Flexibility You Require
| 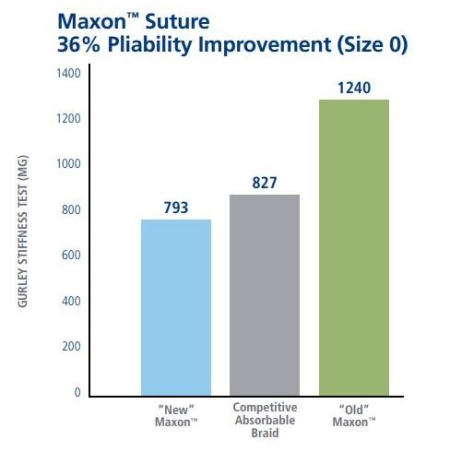 |
Maxon Is Stronger Than The Competition During The Critical Wound Healing Period
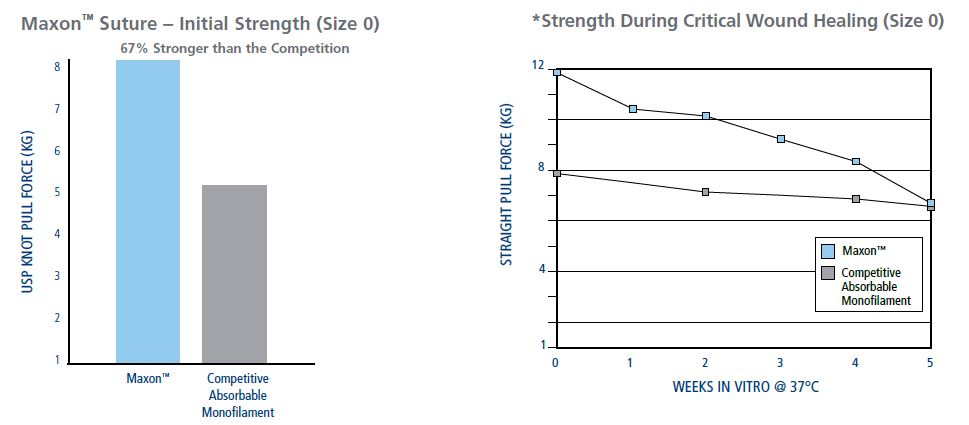
Frequent Uses
General soft tissue approximation including fascia closure, Orthopedic surgery, Pediatric Cardiovascular surgery and patients with compromised wound healing.
Indications
Maxon and Maxon CV synthetic absorbable sutures are indicated for use in general soft tissue approximation and/or ligation, including use in pediatric cardiovascular tissue, where growth is expected to occur, and in peripheral vascular surgery. Maxon and Maxon CV synthetic absorbable sutures are not indicated for use in adult cardiovascular tissue, ophthalmic surgery, microsurgery and neural tissues.
Contraindications
The use of this suture is contraindicated in patients with known sensitivities or allergies to its components. This suture, being absorbable, should not be used where extended approximation of tissue is required or for fixation of permanent cardiovascular prostheses or synthetic grafts.
Composition
Polyglyconate Copolymer of Glycolic Acid and Trimethylene Carbonate
Tensile Strength
80% of initial tensile strength retained at 1 week, 75% at 2 weeks, 65% at 3 weeks, 50% at 4 weeks and 25% at 6 weeks post implant
- 2 weeks: 70%USP
- 4 weeks: 50%USP
- 6 weeks: 25%USP
Device Characteristics
| What MRI safety information does the labeling contain? | Labeling does not contain MRI Safety Information |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437): | No |
| Device labeled as "Not made with natural rubber latex": | No |
| For Single-Use: | Yes |
| Prescription Use (Rx): | Yes |
| Over the Counter (OTC): | No |
| Kit: | No |
| Combination Product: | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P): | No |

Covidien #SMM5044, Suture Maxon Mono Clr P12 3-0 18" 36/BX
$491.44 per BOX

Covidien #SMM5042, Suture Maxon Mono Clr P12 5-0 18" 36/BX
$491.44 per BOX

Covidien #8886660831, Suture Maxon Mono Clr C13 4-0 18" 36/BX
$331.20 per BOX

Covidien #SMM5536, Suture Maxon Mono Clr P13 4-0 18" 36/BX
$750.00 per BOX


